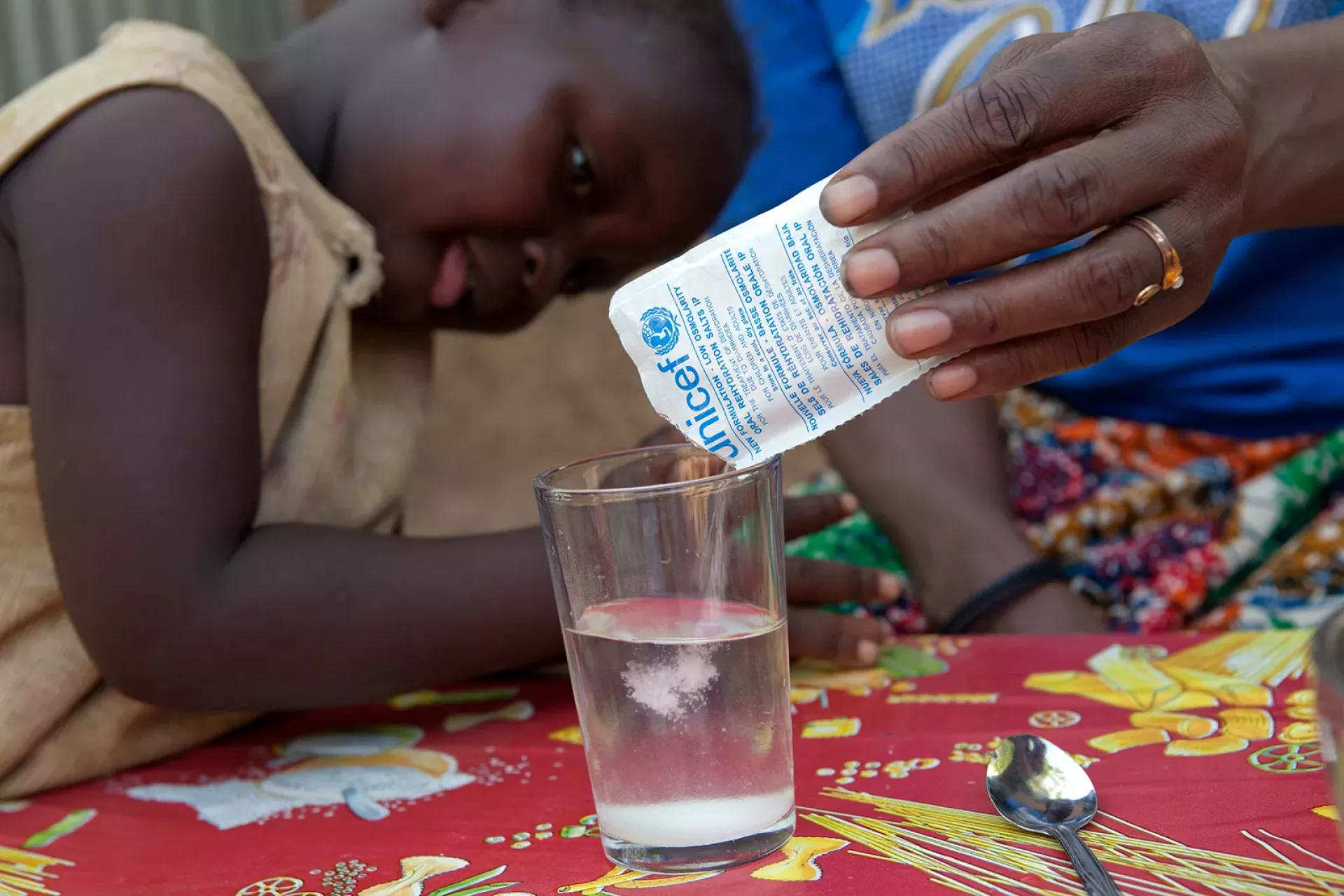
India bans ‘ORS’ label on non-WHO approved beverages, supplements

The Union government has instructed that only products approved by the World Health Organization may be labelled as Oral Rehydration Salts (ORS), reversing earlier permissions that allowed “ORS” to appear in product names with caveats.
In a directive issued October 15, the Food Safety and Standards Authority of India (FSSAI) said the use of “ORS”, even as a prefix or suffix in a trademark or brand name, would no longer be permitted for any food product, whether fruit-based, non-carbonated, or ready-to-drink. The decision takes immediate effect.
The order withdraws earlier permissions granted in July 2022 and February 2024, which had allowed companies to use the term “ORS” alongside a disclaimer stating that the product was not a WHO-recommended formula. The FSSAI stated that these relaxations had been found to cause “false, deceptive, ambiguous, and erroneous” labelling.
The regulator noted that the term “ORS” carries strong public trust and its misuse could “mislead consumers by way of false and deceptive claims,” particularly in products not medically intended to treat dehydration.
The order reaffirms that a previous directive, dated April 8, 2022, concerning the misleading advertising and marketing of ORS substitutes, remains in effect.
FSSAI’s move comes amid growing concern that the misuse or misbranding of ORS could mislead consumers. ORS is a simple solution of salts and sugar in water designed to treat dehydration caused by diarrhoea, heat stroke or other conditions involving fluid loss.
Hyderabad-based Paediatrician Dr Sivaranjani Santosh, who has campaigned against mislabelled ORS products, praised the decision: “No one can use ORS on their label unless it is a WHO-recommended formula, and no one can sell it right from today.”
Health activists and doctors lauded Dr Santosh's eight-year-long persistent campaign against sugar-rich beverages falsely marketed as ORS.